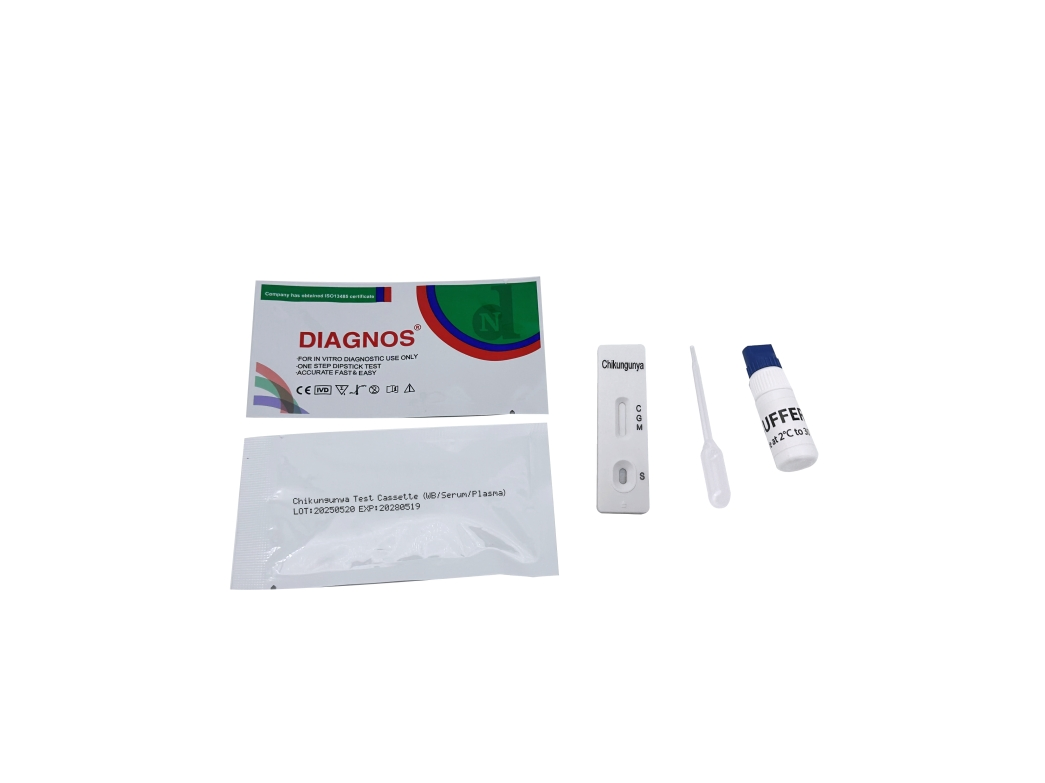
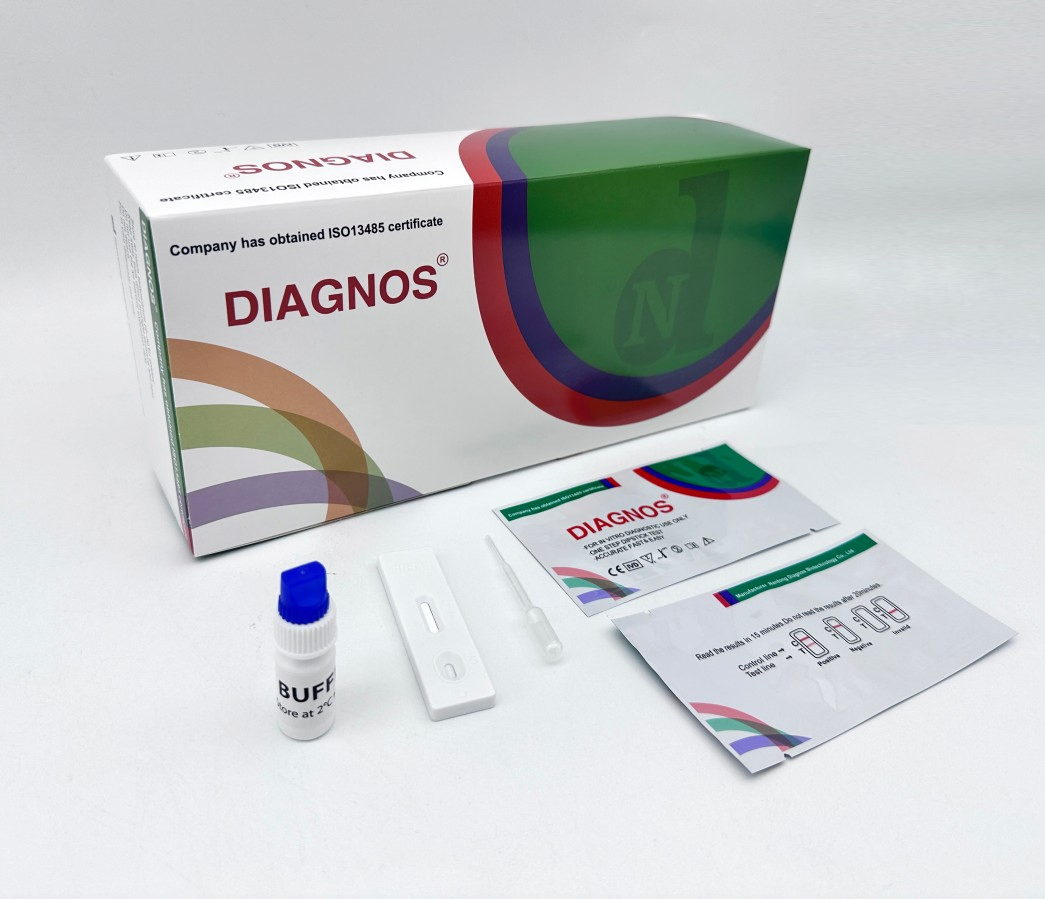
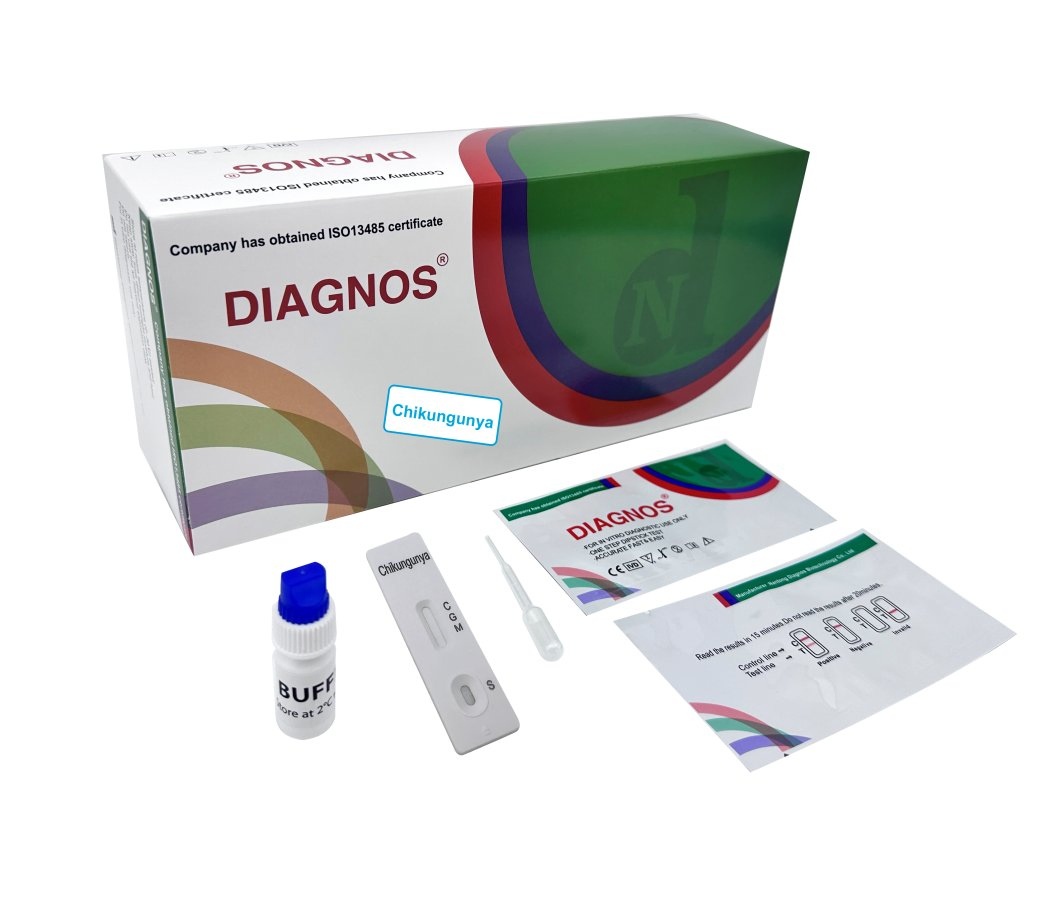










| Model NO. | DNSCHIK | Specimen | Whole Blood/Serum/Plasma |
| Quality Guarantee | 2 Years | Storage | 2°C - 30°C, Cool Dark Dry place |
| Logo Printing | OEM Available | Customized | Available |
| Package | Standard Packing | Specification | 1/25/40 Test |
| Trademark | Diagnos | Origin | China |
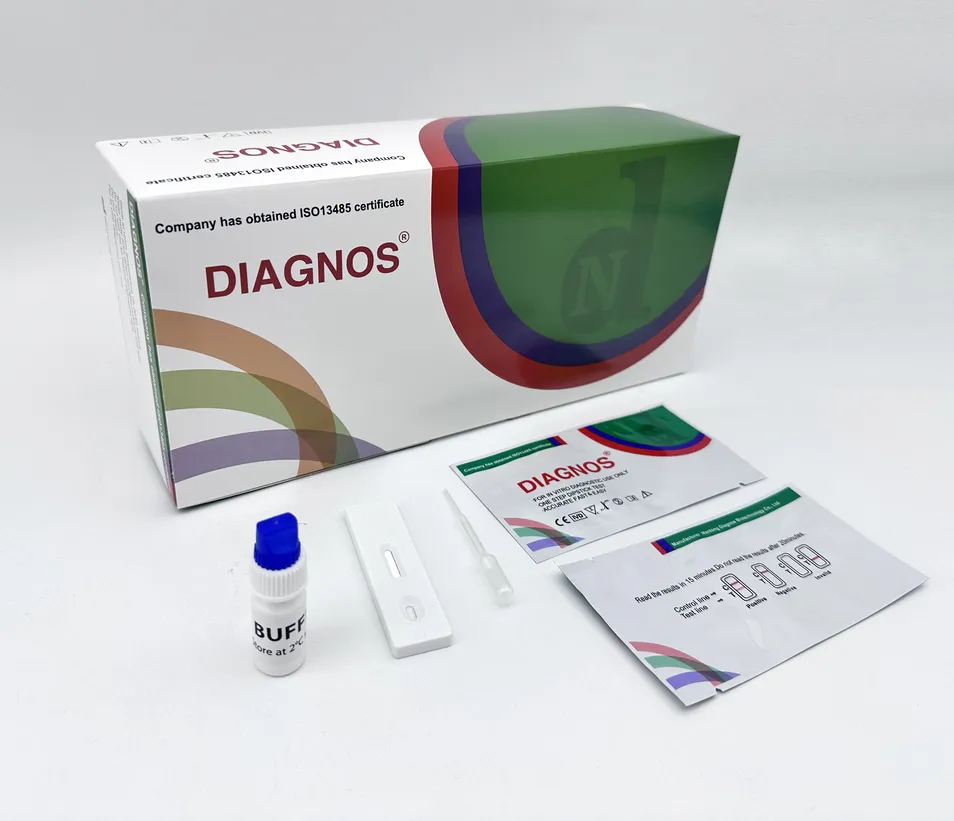
| Model | Chikungunya IgG/IgM Test | Specimen | Whole Blood/Serum/Plasma |
| Packing | 40 pcs/box, 2000 pcs/carton | MOQ | 2000 pcs |
| Certificate | ISO & CE | OEM | Supported |
| Shelf life | 3 years | Production capacity | 20,000,000 pcs |
Chikungunya IgG/IgM Rapid Test is a qualitative test for the detection of IgM and IgG antibodies to chikungunya virus in human serum, plasma. This test is for in-vitro diagnostic use only.
Chikungunya fever is mainly transmitted by Aedes aegypti and Aedes albopictus. The disease is prevalent in tropical and subtropical regions, and is an arboviral disease with the widest distribution, the largest number of morbidities and greater harm in the world.
Following Chikungunya virus infection, IgM antibodies appear approximately 5 days after infection, increase within 1-3 weeks, and persist for 6-9 days. IgG antibodies generally appear until day 14 after primary infection and on day 2 of secondary infection.


Established in 2010, the company is a technology-based enterprise specializing in the research and development, production, sales and service of in vitro rapid diagnostic reagents. Utilizing monoclonal antibody technology, molecular immunology technology, nanotechnology and microelectronics technology, we accelerate product development to launch rapid, sensitive, and accurate diagnostic series.
Adhering to the business philosophy of "quality, reputation, service and sincerity," all product performances and indicators meet strict industry standards. We focus on independent R&D and standardized management to provide innovative healthcare solutions.



