Industry-Leading Sterile Packs for Precision Medical Operations

Surgical Pack Medical Disposable Basic Surgical Procedure Kits Sterile Surgical Drape
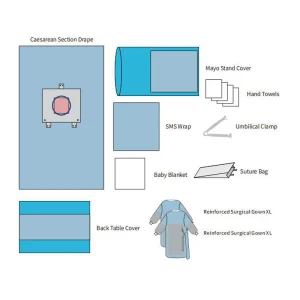
Eo Sterile Universal Surgical Drape Kit Angiography/Cardiovascular Surgical Procedure Pack

OEM Disposable Surgical Pack/Drape Sterile Surgical Kit for Surgery Operation

Sterile Surgical Pack Disposable Examination Drape for Orthopedics and Total Joint Procedures
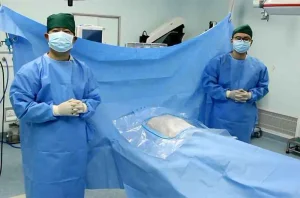
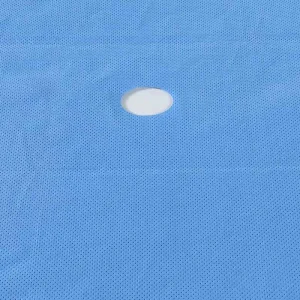
CE ISO Disposable Medical Surgical Universal Pack Set Operation Drape


High-Quality Disposable Transparent Surgical Film Adhesive Wound Dressing Non-Woven Surgical Drape
The global market for Surgical Prep Pads and sterile disposables has seen an unprecedented surge. With the increasing complexity of surgical procedures and the heightened awareness of Surgical Site Infections (SSIs), medical facilities worldwide are transitioning from traditional reusable textiles to high-performance, single-use medical consumables.
Currently, the demand is driven by the rise in outpatient surgeries and the expansion of healthcare infrastructure in emerging economies. Manufacturers are now pressured to provide not just a product, but a comprehensive infection prevention solution that meets stringent FDA and MDR (Medical Device Regulation) standards.
Technology is redefining pre-operative care. We are witnessing a shift toward "Smart Prep" materials—non-woven fabrics that offer superior fluid absorption while maintaining a breathable barrier. Innovations in antiseptic chemistry, such as advanced saturation levels for alcohol or CHG (Chlorhexidine Gluconate), are becoming the new industry benchmark.
Sustainability is another key trend. As a leading manufacturer, we are exploring eco-friendly materials that reduce the environmental footprint of medical waste without compromising sterility or patient safety.
For global procurement officers, the focus has shifted from price alone to Supply Chain Resilience and quality consistency. International buyers require manufacturers who can provide comprehensive documentation, including sterilization certificates, biocompatibility reports, and ISO 13485 compliance.
Localization is also key; the ability to customize surgical packs (OEM/ODM) to meet the specific cultural and clinical needs of different regions—such as specialized drape sizes for orthopedic vs. dental procedures—is a top priority for modern distributors.
Shanghai SwellMed Medical Co., Ltd. is a globally recognized manufacturer of high-quality single-use sterile medical devices, with a strong focus on infection control and surgical safety.
We support OEM & ODM customization for healthcare providers, distributors, and brand owners worldwide. Our core strength lies in seamlessly integrating cutting-edge R&D with practical clinical application. We possess deep domain expertise and hold numerous patents for our innovative technologies.
Our products, backed by robust clinical data, are designed with a user-centric approach, ensuring reliability, ease of use, and improved patient outcomes. Furthermore, our global compliance certifications (e.g., FDA, CE) and dedicated professional support team guarantee trust and partnership at every stage.


Our medical devices have demonstrated exceptional clinical value, supporting over 50,000 successful procedures worldwide. Sales have grown 200% in three years, driven by surgeon trust and proven results in enhancing surgical precision and reducing recovery times.
This performance underscores our role as a key innovator in the medical field. From metropolitan hospitals in Europe to field clinics in Southeast Asia, SwellMed products are synonymous with safety and excellence. We continue to invest in automated production lines to ensure that every surgical prep pad meets the same high standard of sterility and performance.
Tailored Solutions for Diverse Medical Environments
High-saturation prep pads and universal drapes provide a sterile field for abdominal, thoracic, and emergency surgeries, ensuring fluid management and skin antisepsis are handled with professional precision.
Specific dental implant kits designed for the restricted field of oral surgery. These kits include specialized drapes that prevent contamination while allowing the surgeon maximum visibility and movement.
Orthopedic surgeries require heavy-duty fluid collection and maximum barrier protection. Our sterile packs for total joint procedures are engineered to withstand the rigors of long, complex bone surgeries.
Precision-focused kits for catheterization labs and angiography suites, offering femoral or radial access points with integrated prep pads for immediate site cleaning.
The Strategic Advantage of Modernized Production Hubs
1. Advanced Infrastructure & Scale: China has invested heavily in "Medical Valley" industrial zones that consolidate the entire supply chain—from raw non-woven fabric production to EO sterilization facilities. This integration reduces lead times and ensures competitive pricing for bulk procurement.
2. R&D and Customization (OEM/ODM): Unlike traditional suppliers, modern Chinese factories like SwellMed are R&D-driven. We can rapidly prototype new pack configurations or prep pad materials based on clinical feedback from international partners.
3. Rigorous Quality Compliance: Today's leading China manufacturers operate under strict international standards. With CE (MDR), FDA, and ISO 13485 certifications, our surgical products are welcomed in the most regulated markets including the USA and the EU.
4. Logistic Efficiency: Located in Shanghai, we leverage one of the world's most efficient logistics hubs, providing seamless sea, air, and rail transport options to ensure your medical inventory is never at risk of stockouts.
Reliable Protective Gear and Sterile Solutions for Every Procedure
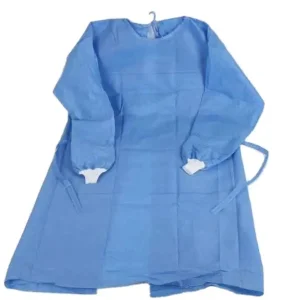
Non-Reinforced Surgical Gown with Towel Cypress X-Large Blue Sterile AAMI Level 3 Disposable


Medical Disposable Operation Surgery Universal Pack Surgical Procedure Packs


AAMI Level 2 Medical Disposable Surgical Isolation Gown OEM Customized Protective Gown Surgical Gown

Disposable Medical Sterile Customized Surgery Pack for Different Surgical Procedures

Disposable Medical Complete Sterile Surgery Pack for Surgical Procedures
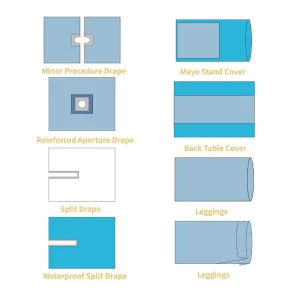
Customizable & Pre-Assembled Sterile Drape Packs for Surgical Procedures
As the healthcare landscape evolves, the role of a Surgical Prep Pad Manufacturer extends beyond simple production. It involves deep clinical expertise. Our team at Shanghai SwellMed works closely with frontline surgeons to understand the micro-challenges of the operating room. Whether it is the tear-resistance of a drape or the rapid-dry time of a prep pad, every detail is engineered to reduce the risk of cross-contamination.
Our commitment to E-E-A-T principles means that we don't just sell products; we provide documented assurance. Our clinical data shows that using our high-grade sterile packs can contribute to a significant decrease in post-operative complications. This data-driven approach is what has led to our 200% growth and the trust of over 50,000 procedures globally.
In the competitive world of medical supplies, the difference lies in reliability. A single failed sterile barrier can have catastrophic consequences. This is why we implement a multi-stage quality control process, including raw material testing, cleanroom environment monitoring, and post-sterilization validation. When you choose SwellMed, you are choosing a partner dedicated to the preservation of life through superior infection control.
Looking forward, we are expanding our R&D into bio-compatible dressings and advanced surgical films. We invite global distributors and healthcare institutions to explore our manufacturing capabilities and join us in setting a new standard for surgical excellence. Together, we can create a safer clinical environment for patients everywhere.
By focusing on automation and AI-driven quality checks, our factory ensures that every batch of surgical prep pads is identical in quality. This consistency is vital for large-scale hospital groups that manage hundreds of thousands of procedures annually and require a predictable supply of high-performance medical consumables.
In summary, the transition to high-quality disposable surgical products is not just a trend—it is a global shift toward safer, more efficient healthcare. As a top China manufacturer, Shanghai SwellMed is at the forefront of this movement, ready to serve the world with expertise, professionalism, and unwavering reliability.