 Surgical Pack Medical Disposable Basic Surgical Procedure Kits Sterile Surgical Drape
Surgical Pack Medical Disposable Basic Surgical Procedure Kits Sterile Surgical Drape
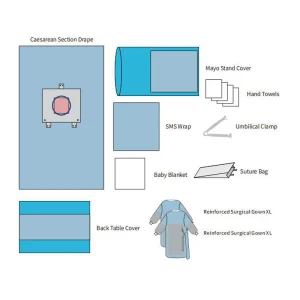 Eo Sterile Universal Surgical Drape Kit Angiography/Cardiovascular Surgical Procedure Pack
Eo Sterile Universal Surgical Drape Kit Angiography/Cardiovascular Surgical Procedure Pack
 OEM Disposable Surgical Pack/Drape Sterile Surgical Kit for Surgery Operation
OEM Disposable Surgical Pack/Drape Sterile Surgical Kit for Surgery Operation
 Sterile Surgical Pack Disposable Examination Drape for Orthopedics and Total Joint Procedures
Sterile Surgical Pack Disposable Examination Drape for Orthopedics and Total Joint Procedures
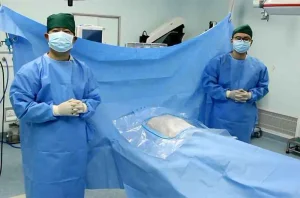 Disposable Sterile Surgical Pack Kit Dental Implant Drape
Disposable Sterile Surgical Pack Kit Dental Implant Drape
 CE ISO Disposable Medical Surgical Universal Pack Set Operation Drape
CE ISO Disposable Medical Surgical Universal Pack Set Operation Drape
 Disposable Sterile Universal Pack Surgical Drape
Disposable Sterile Universal Pack Surgical Drape
 OEM Medical Consumables Wound Care/Circumcision Procedure Pack/Basic Sterile Surgical Drape Set Pack with CE13485
OEM Medical Consumables Wound Care/Circumcision Procedure Pack/Basic Sterile Surgical Drape Set Pack with CE13485
In the modern healthcare era, surgical site infections (SSIs) remain a critical challenge for hospitals worldwide. As the first line of defense, Surgical Hand Sanitizers have evolved from simple soaps to sophisticated pharmacological formulations designed to ensure persistent antimicrobial activity.
The global surgical hand disinfectant market is projected to grow at a CAGR of 7.2% through 2030. This surge is driven by the increasing volume of surgical procedures and a heightened awareness of hospital-acquired infections (HAIs) in emerging economies.
Compliance with international standards like EN 1500, EN 12791, and ASTM E1174 is no longer optional. Industrial-grade sanitizers must now provide a 3-hour persistent effect to protect surgical teams during long-duration operations.
There is a significant shift toward "green chemistry." Modern factories are now prioritizing biodegradable ingredients and recyclable packaging to meet the ESG (Environmental, Social, and Governance) goals of global healthcare groups.
The industry is moving beyond the liquid in the bottle. We are seeing the integration of AI and IoT in hand hygiene monitoring. Smart dispensers now track compliance rates in real-time, providing data-driven insights to infection control departments.
Furthermore, the "Waterless Scrub" revolution is gaining momentum. Alcohol-based hand rubs (ABHR) are replacing traditional water-aided scrubbing, reducing skin irritation for surgeons and saving thousands of gallons of sterile water per hospital annually.

Large-scale procurement focuses on supply chain resilience and bulk pricing. We provide 5L and 10L refills compatible with centralized dispensing systems used in Tier-1 hospitals.
In localized scenarios like field hospitals or disaster relief, waterless surgical sanitizers are indispensable. Our portable, high-concentration units ensure safety where infrastructure is lacking.
Dental and outpatient surgery centers require rapid-turnover hygiene. Our fast-drying, non-sticky formulas allow for quick gloving and high patient throughput.
As a premier Chinese exporter, we combine massive production capacity with rigorous quality control, offering a competitive edge that Western manufacturers often struggle to match.
Compliance with ISO 13485
Global Export Countries
R&D Technical Support
Custom Branding Options
Leveraging China's integrated chemical supply chain, we provide medical-grade ethanol and isopropanol solutions at a fraction of the cost of local distributors.
Our lab focuses on the synergy between biocides and skin protectants, ensuring that high-frequency use does not lead to contact dermatitis among medical staff.
From hazardous material handling to global shipping, our export team ensures that your surgical sanitizers arrive on time, with all regulatory paperwork cleared.
Shanghai SwellMed Medical Co., Ltd. is a globally recognized manufacturer of high-quality single-use sterile medical devices, with a strong focus on infection control and surgical safety.
We support OEM & ODM customization for healthcare providers, distributors, and brand owners worldwide.
Our core strength lies in seamlessly integrating cutting-edge R&D with practical clinical application. We possess deep domain expertise and hold numerous patents for our innovative technologies. Our products, backed by robust clinical data, are designed with a user-centric approach, ensuring reliability, ease of use, and improved patient outcomes. Furthermore, our global compliance certifications (e.g., FDA, CE) and dedicated professional support team guarantee trust and partnership at every stage.
Our medical devices have demonstrated exceptional clinical value, supporting over 50,000 successful procedures worldwide. Sales have grown 200% in three years, driven by surgeon trust and proven results in enhancing surgical precision and reducing recovery times. This performance underscores our role as a key innovator in the medical field.

 Medical Disposable Operation Surgery Universal Pack Surgical Procedure Packs
Medical Disposable Operation Surgery Universal Pack Surgical Procedure Packs
 High-Quality Disposable Transparent Surgical Film Adhesive Wound Dressing Non-Woven Surgical Drape
High-Quality Disposable Transparent Surgical Film Adhesive Wound Dressing Non-Woven Surgical Drape
 China High Quality Disposable Medical Surgical Pack Universal Sterile Drape and Isolation Kit for Dental Dialysis and C-Section Procedures
China High Quality Disposable Medical Surgical Pack Universal Sterile Drape and Isolation Kit for Dental Dialysis and C-Section Procedures
 Disposable Medical Sterile Customized Surgery Pack for Different Surgical Procedures
Disposable Medical Sterile Customized Surgery Pack for Different Surgical Procedures
 Disposable Medical Complete Sterile Surgery Pack for Surgical Procedures
Disposable Medical Complete Sterile Surgery Pack for Surgical Procedures
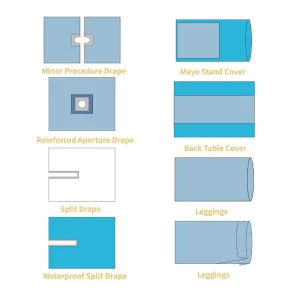 Customizable & Pre-Assembled Sterile Drape Packs for Surgical Procedures
Customizable & Pre-Assembled Sterile Drape Packs for Surgical Procedures
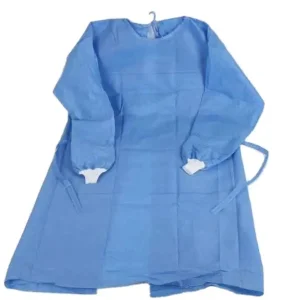 Non-Reinforced Surgical Gown with Towel Cypress X-Large Blue Sterile AAMI Level 3 Disposable
Non-Reinforced Surgical Gown with Towel Cypress X-Large Blue Sterile AAMI Level 3 Disposable
 AAMI Level 2 Medical Disposable Surgical Isolation Gown OEM Customized Protective Gown Surgical Gown
AAMI Level 2 Medical Disposable Surgical Isolation Gown OEM Customized Protective Gown Surgical Gown
The efficacy of Surgical Hand Sanitizers is paramount in the operating theater. Unlike standard consumer hand rubs, surgical-grade disinfectants must meet the stringent requirements of the "surgical hand rub" protocol. This involves a multi-stage application process that ensures the reduction of transient flora and the suppression of resident flora for the duration of the surgery. Our factory in China utilizes advanced distillation and filtration processes to ensure the purity of our active ingredients, exceeding the USP and BP standards for medical alcohols.
In the context of global health security, the diversification of supply chains has become a strategic priority for many nations. By partnering with a high-capacity Chinese manufacturer like SwellMed, international buyers secure a reliable stream of essential infection control products. We understand the nuances of different markets—from the specific labeling requirements of the EU's BPR (Biocidal Products Regulation) to the FDA's OTC monograph system in the United States.
Technological integration is the next frontier. We are currently developing "Smart Formulations" that change color slightly upon reaching the required contact time, providing a visual cue to the surgeon that antisepsis is complete. This type of innovation is what sets our factory apart as a leader in the medical consumables industry. Our commitment to E-E-A-T principles means that every bottle of sanitizer we export is backed by rigorous laboratory testing, real-world clinical feedback, and a transparent manufacturing process.
Furthermore, the role of surgical hand sanitizers extends into the veterinary, pharmaceutical cleanroom, and laboratory sectors. Each of these localized application scenarios requires a slightly different approach to formulation—whether it's the removal of fragrances for lab environments or the addition of extra skin conditioners for veterinary surgeons who perform multiple procedures daily. Our OEM capabilities allow us to tailor these formulations to your specific market needs, ensuring that your brand stands for quality and safety.
As we look to the future, the focus on "One Health" and the prevention of antimicrobial resistance (AMR) will drive the development of new biocidal agents. Our R&D team is already exploring non-alcohol based persistent antimicrobial agents that provide long-lasting protection without the flammability risks associated with high-concentration alcohols. This forward-thinking approach ensures that our partners are always at the cutting edge of surgical safety technology.